Written by Tim Peterson, assistant professor, Washington University School of Medicine
This post is a lay introduction on how molecular biology is increasingly being used to make diagnoses and treatments better tailored to individual patients.
The Definitions
For those who may not be familiar with molecular biology, there are three main types of molecules in our bodies – DNA, RNA, protein – that microscopically orchestrate everything about us.1 They coordinate the vastly complex processes that allow us to eat, breathe, sleep, etc. Our DNA can be thought of an instruction manual. It has rules to produce RNAs that have other rules to make proteins. The rules are what people know of as genes. Proteins do much of the physical work in our cells. They are quite literally little biological machines. The above is albeit an over-simplification, but it is a reasonable framework for this piece.
Personalized medicine and precision medicine are synonyms.2 They mean treatments that are individualized. Precision medicine is probably a better term for what many people refer to when they say, “personalized medicine” as I’ll get into below. Pharmacogenetics and pharmacogenomics are related terms to precision medicine. They refer to how a drug interacts with a gene (genetics) or genes (genomics). By “interact”, it’s the protein made from a gene that is actually, in most cases, what physically interacts with a drug. “-ome” is a suffix that biologists have latched onto to refer to a collection of molecules. Because people’s genes differ in their DNA instruction manuals, even surprisingly slightly, they respond to drugs differently.
What’s Old Is New Again
Doctors have been doing personalized medicine based on our molecular biology for years. By definition, medical care is personalized. Doctors prescribe medicine to a patient based on what’s specifically wrong with them. For example, they only give a patient a drug “X” if their lab results show altered levels of “Y”. So, if a Type I Diabetic patient’s glucose is too high, then they get insulin to bring the glucose levels back down.3 It’s individualized in the sense that not everyone has high levels of glucose.
What’s different now is it’s getting more precise. Now, the doctor might give different amounts of drug “X” or they might give drug “Z” if the patient has altered levels of “Y”. Deciding how much “X” to give or deciding between “X” and “Z” has traditionally been based on clinical results and physician experience. Increasingly, molecular information is being incorporated into the decision making.
New Molecular Diagnostics In the Clinic
A major advance is that doctors are now able to analyze a patient’s DNA and make treatment decisions based on it. An example is the BRCA mutations for breast cancer.4 Most of these tests are in the early stages, but there is a lot of interest. There are also newer tests looking at proteins and RNAs, but DNA testing has arguably received the most focus in the precision medicine field.
We have roughly 20,000 genes that make proteins according to the “central dogma” of biology: DNA -> RNA -> protein I mentioned above. Proteins can interact with a drug in two general ways. They can influence drug pharmacokinetics or pharmacodynamics.5
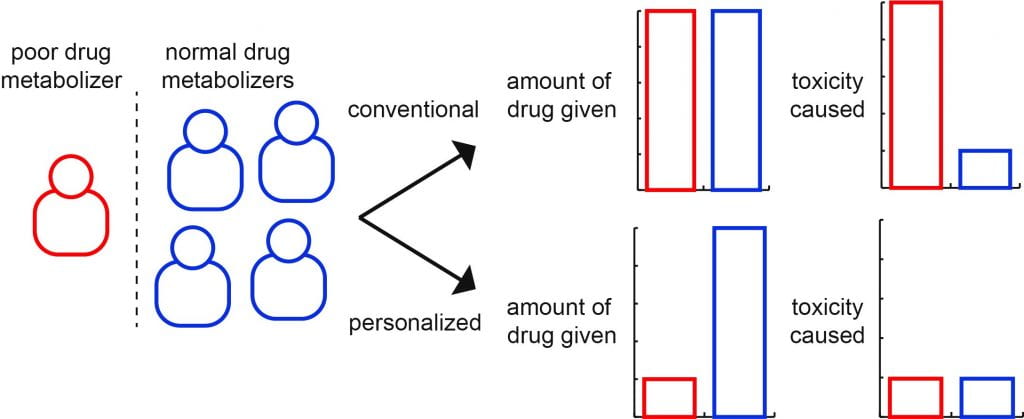
Pharmacokinetics refers to what our bodies do to a drug. For example, does one of our proteins aid in the degradation or excretion of the drug from the body? There are proteins, namely the CYP450 family, that are abundant in the liver that influence the pharmacokinetics of many different types of drugs.6 They do such things as chemically modify drugs to increase their water solubility causing them to be excreted into the urine more readily. People can differ significantly in their CYP450 genes. How this translates to precision medicine is that a doctor can look at the DNA that codes for the CYP450 proteins and see whether they might metabolize a drug better or worse. Most people metabolize drugs similarly, but the dangerous scenario is if a person doesn’t metabolize a drug well and it builds up to toxic levels (see figure above). This is where precision medicine can save lives.
Pharmacodynamics refers to how a drug affects our body. For example, does it inhibit a protein’s activity? This inhibition can often be the drug’s desirable, therapeutic effects, but it can also be an unwanted side effect. An example is the bisphosphonates, which are drugs for osteoporosis (bone loss).7 The bisphosphonates increase bone density, but in rare cases they can cause fractures. Pharmacodynamics like pharmacokinetics are getting more precise as we learn more about what our genes normally do. With bisphosphonates, we now know that variation in the gene, GGPS1, may be important for people who get fractures from taking bisphosphonates.8 However, there are fewer pharmacodynamics-based diagnostics because in general, unlike with the CYP450-based diagnostics, a different diagnostic tool has to be developed for each drug because each drug has a different therapeutic target protein.
Newer, More Precise Treatments
It’s not just diagnostics that are getting more precise. Treatments are too. Again, the DNA holds lots of the answers to these improvements. Because we can know a patient’s DNA sequence (and the protein it codes for) we can develop drugs specifically for that person. This is particularly important in cancer because the DNA gets mutated the same way in specific cancers every time. Therefore, we can make drugs that will be precisely effective for those cancers.9 An example is the Herceptin drug for breast cancers that over-express the protein for the HER2 gene.10
Democratizing Molecular Medicine
With all the above being said, we still need special (expensive) equipment to acquire and make use of molecular information. Yet, one can imagine a democratization of molecular biology like we’ve witnessed with the personal computer. Some companies, such as 23AndMe, are even promising this to people now. If the technology is useful and accurate, it will go mainstream.
The real challenges to using this new molecular knowledge, particularly in using our DNA, are legal and business and not science.11 As a society, we only accept technology when we are “ready.” Our DNA is our personal information and we can’t change it. There are legitimate fears about other people and powerful organizations having access to it. How do we protect patients and abide by privacy laws such as HIPAA?12 How do we prevent people from getting discriminated against based on having the “wrong” DNA?
While there’s no organization on earth that everyone unilaterally trusts to manage the world’s DNA, Apple seems to have a decent shot at leading the sea change. As an example, they’ve recently announced a partnership with health systems such as Johns Hopkins University to bring medical records into their Health app.13 It’s an exciting development. But it’s also the adage, “with great power comes great responsibility.” Through these partnerships and other initiatives at WashU14 and elsewhere, we must be diligent and work together to use the molecules that make us for our own good.
Conflict of Interest statement: Tim Peterson owns Apple stock, but has no relationship with 23AndMe. This is an opinion piece and not financial, legal or medical advice.
References
- https://simple.wikipedia.org/wiki/Central_dogma_of_molecular_biology.
- https://ghr.nlm.nih.gov/primer/precisionmedicine/precisionvspersonalized.
- https://en.wikipedia.org/wiki/Diabetes_mellitus_type_1.
- https://www.cancer.gov/about-cancer/causes-prevention/genetics/brca-fact-sheet.
- https://en.wikipedia.org/wiki/Pharmacodynamics.
- https://ghr.nlm.nih.gov/primer/genefamily/cytochromep450.
- https://en.wikipedia.org/wiki/Bisphosphonate.
- http://www.nejm.org/doi/full/10.1056/NEJMc1612804.
- https://www.cancer.gov/about-cancer/treatment/types/precision-medicine.
- http://www.breastcancer.org/treatment/targeted_therapies/herceptin/how_works.
- https://precisionmedicine.duke.edu/ethical-social-legal-issues/ethicalsocial-policy.
- https://www.hhs.gov/hipaa/index.html.
- https://www.apple.com/newsroom/2018/01/apple-announces-effortless-solution-bringing-health-records-to-iPhone/.
- https://publichealth.wustl.edu/tag/precision-medicine/.